Welcome to the Brown University TGEF!
We provide one-stop shop services for generating genetically modified (GM) mouse or rat models, covering project consultation, novel model design, reagents, model production, germline transmission, and genotyping. The deliverables of novel models are F1 heterozygous mice or rats, along with their genotyping analysis protocol.
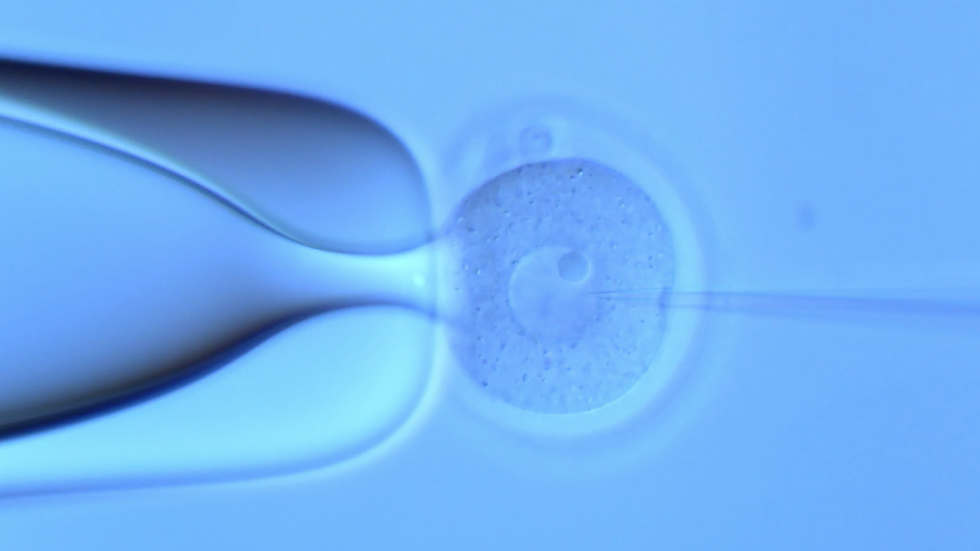
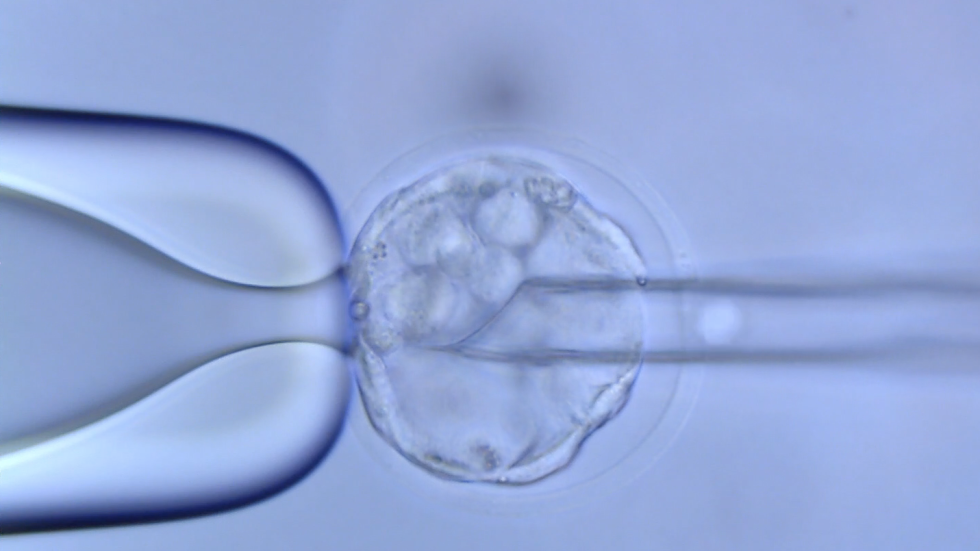
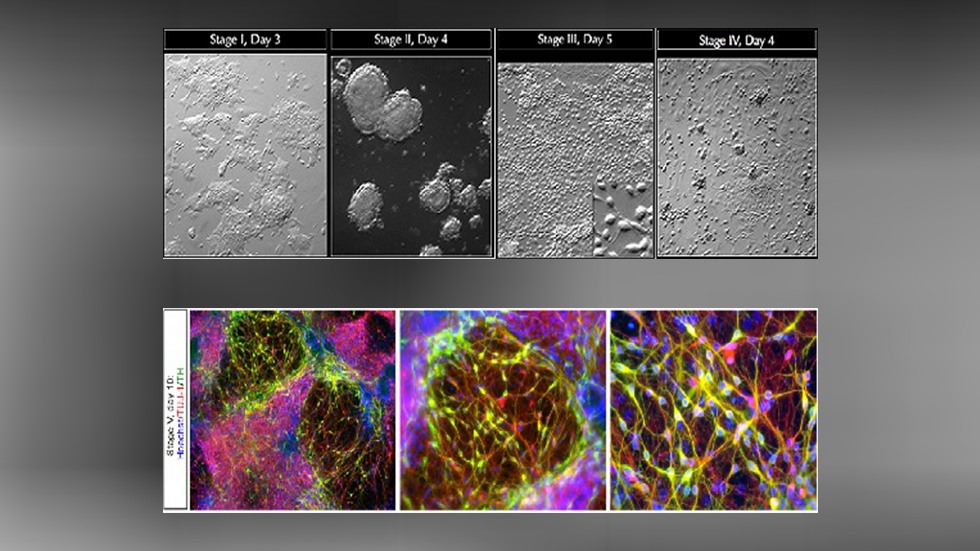
The cutting-edge technologies of the CRISPR/Cas platform are primarily employed at the TGEF to accomplish the vast majority of projects in a cost-effective, fast, and high-quality manner. The conventional ES cell gene-targeting system is employed as a complementary approach to overcome the limitations of the CRISPR/Cas system. Routine services include genotype analysis, sperm or embryo cryopreservation and storage, mouse or rat (frozen embryos) line rederivation, in vitro fertilization (IVF). Other services, such as mouse vasectomy, embryo transfer, colony scale-up, and intracytoplasmic sperm injection (ICSI) are also available. New services requiring TGEF resources can be created after consulting with us.